When drawing the number seven don’t forget about the seventh element, hydrogen. Shown in the illustration, if you take your finger and draw the number seven, starting at nitrogen, you will see that six of the diatomic elements are arranged in the shape of a number seven. 
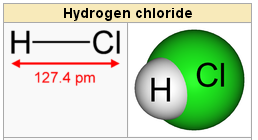
If there is ever a need to identify these diatomic elements on the periodic table, they can be spotted by remembering the ‘seven rule’ as well. One great way to remember which atoms form the lucky seven diatomic elements is to think of the following mnemonic device: I Bring Cookies For Our New Home. Rather, these atoms will always be paired together because they need to pool resources to have enough electrons. That is, you will never find a nitrogen or fluorine atom, for example, hanging out solo. Other elements exist as moleculesfor example, sulfur normally exists as an eight-atom molecule, S 8, while phosphorus exists as a four-atom molecule, P 4 (Figure 1 ). However, with that higher temperature, they will also exist as gases.ĭiatomic elements are special as the atoms that form it do not like to be alone. 1: The element symbols for Bromine, Iodine, Nitrogen, Chlorine, Hydrogen, Oxygen and Fluorine spelling out 'BrINClHOF' a handy mneumonic for memorizing the diatomic elements. At room temperature, bromine and iodine commonly exist in the liquid form. If the temperature is raised slightly higher, two additional elements will be present: bromine and iodine. As an example, the diatomic element oxygen has a formula of O2, meaning there are two separate oxygen atoms present.Īt room temperature, there are five diatomic elements, all of which exist in the gas form: hydrogen, nitrogen, oxygen, fluorine, and chlorine. Every molecule has its own unique molecular formula, and for diatomic elements, their formula regularly contains an addendum of 2, which performs two atoms in its arrangement. If we notice at the prefix ‘di-‘ in the word diatomic, it comes from the Greek origin of ‘two.’ One major way to know you are functioning with a diatomic element is to consider its formula. All of the elements in this noble group are gases. This oxygen, O2, and nitrogen, N2, have something in common: both are called diatomic elements.Īs pure components, diatomic elements are seven particular molecules that are composed of two atoms. To be better precise, nitrogen makes up 78% of the space while oxygen makes up 21%. There are major segments that make up the earth’s atmosphere: nitrogen, oxygen, and much a tad sample of argon. There are seven pure elements that form diatomic Elements. Diatomic compounds consist of two different elements. Many compounds are diatomic, such as HCl, NaCl, and KBr. Other elements also exist naturally as diatomic molecules a molecule with only two ato ms (Table 5.2. For example, hydrogen and oxygen exist as two-atom molecules. Some elements exist naturally as molecules. In contrast, monatomic elements consist of single atoms. A molecule, however, is composed of more than one atom. Writing Chemical Formulas of Diatomic Molecules For a covalent molecule, the information represented in its chemical formula must be a direct reflection of its Lewis structure. Write the correct formulas for all compounds.Diatomic Elements consist of two atoms bonded together. Because this molecule only contains two atoms of the same element, it is classified as a homonuclear diatomic molecule.This will help you know which symbols go on each side of the arrow and where the \( \) signs go. To turn word equations into symbolic equations, we need to follow the given steps: The word equation for this reaction might read something like "solid copper reacts with an aqueous solution of silver nitrate to produce a solution of copper (II) nitrate with solid silver."
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
